| In the staggered conformation (see table below), the ethane molecule has an S6 rotation axis. |
Click for larger image | |
| Using the images in the table, click on the H atom in the product which should be coloured red given the operations performed (Note — rotations are clockwise) about the S6 axis in C2H6. | ||
| (You may find this easier to visualize using Newman projections. Remember, all the H atoms are identical - one has been coloured red merely for convenience!) |
| Operations | Clickable Image | Corresponding Answer |
|---|---|---|
| S6 | 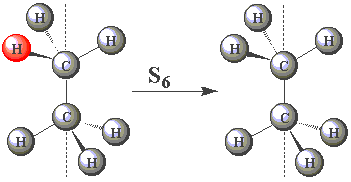 | |
| S62 |  | |
| S63 | 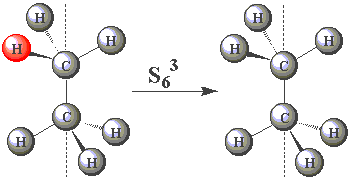 | |
| S64 | 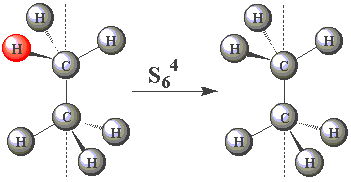 | |
| S65 | 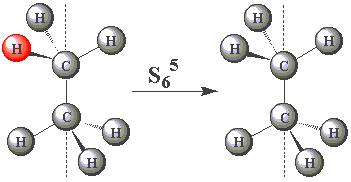 | |
| S66 | 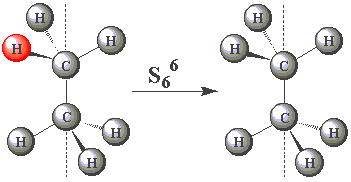 |
Fill in the table below for those S6 operations you think have equivalent symmetry operations generated by other symmetry elements of ethane. Hence, check all the independent symmetry operations we should assign to S6.
In general, if n is even and n/2 is odd, then Snn/2 ≡ i.
| Previous (Exercise 1) | Return to top of page | Next (Exercise 3) |